A three-year-old boy diagnosed before birth with a severe form of spina bifida is able to walk following experimental stem cell surgery performed in utero, according to researchers involved in a small early-stage clinical trial.
During a routine ultrasound at 20 weeks of pregnancy, Tobi Maginnis was found to have myelomeningocele, the most serious form of spina bifida, a condition in which the spinal cord does not close properly during early fetal development. The defect can leave part of the spinal cord exposed and is often associated with paralysis of the lower limbs as well as bowel and bladder dysfunction.
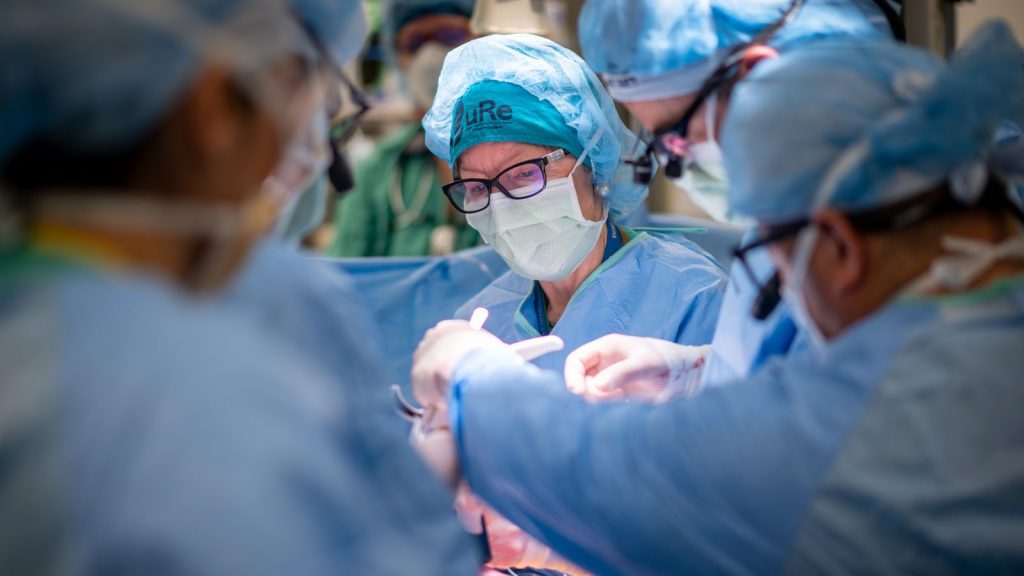
Tobi became one of six fetuses enrolled in a first-of-its-kind clinical trial conducted at University of California Davis Children’s Hospital. The procedure involved transplanting stem cells derived from placental tissue directly onto the exposed portion of the fetal spinal cord during surgery.
At approximately 25 weeks’ gestation, surgeons made a small incision in the mother’s abdomen and uterus to access the fetus. A patch containing the stem cells was placed over the spinal defect before the uterus was closed, allowing the pregnancy to continue. The aim was to encourage tissue repair and improve neurological outcomes beyond what is typically achieved with standard prenatal surgery.
Results from the trial, published in The Lancet, reported that all six babies were born without immediate complications attributed to the stem cell treatment. Researchers did not observe short-term adverse effects related to the procedure.
According to his parents, Tobi is now able to walk, run and jump—an outcome they say exceeded their expectations given the severity of his diagnosis. His father noted that, as only the second human patient to undergo the stem cell–augmented procedure, there had been considerable uncertainty surrounding potential outcomes.
The study was led by Dr Diana Farmer, a fetal surgeon who has previously worked on advancing prenatal interventions for spina bifida. She described the early results as encouraging but emphasised caution, noting that the long-term safety and effectiveness of introducing stem cells into the fetal spinal cord remain under evaluation. Researchers continue to monitor participants for any delayed or unforeseen effects.
Spina bifida affects hundreds of pregnancies each year. In the United Kingdom, approximately 500 babies annually are born with myelomeningocele. While prenatal surgical repair of the spinal defect has become more widely practiced and has been shown to improve some outcomes compared with postnatal surgery, studies indicate that around half of treated children are still unable to walk independently.

The investigators in California hope that adding stem cells to conventional fetal surgery may enhance nerve regeneration and improve mobility and bladder control. A larger follow-up trial involving 35 pregnant women has now begun. Participants’ children will be tracked for several years to assess longer-term neurological function and to determine whether the approach provides measurable advantages over existing surgical techniques.
Researchers stress that the findings are preliminary and that broader clinical data will be needed before the procedure could be considered for wider adoption.



